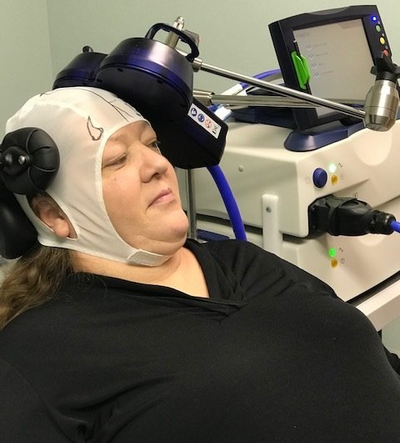
Transcranial Magnetic Stimulation (TMS)
TREATMENTS FOR TRD (TREATMENT RESISTANT DEPRESSION):
Dr. Vik Shukla completed on site TMS training for administering TMS. He attends regular CME activities for TMS online as well as American Psychiatric Association CME meeting for TMS prior to establishing TMS service for his office for the year 2020 and beyond.
What is TMS?
TMS is a FDA approved innovative and non-invasive treatment for TRD (treatment resistant depression) after nonresponse to two or more anti-depressant medications with adequate attempts. TMS does not require surgery or anesthesia, unlike VNS, and monitoring in operating room (as required in ECT: electroconvulsive therapy).
TMS is office based 5 days a week for 6 weeks sit down treatments involving typically 19 minutes (in some cases 3-minute theta burst TMS) per session. Electromagnet wand is placed on accurate brain site mapped personally by physician followed by stated TMS (Repetitive Transcranial Magnetic) for 19 minutes per session. After 6 weeks (30 sessions), maintenance TMS is usually determined and recommended to maintain improvement case by case)
TMS does not require "seizures" of brain at all that is a REQUIREMENT for ECTs that commonly entails short term memory loss. The treatment is safe with none of the side effects of antidepressant medications.
Currently TRD and OCD (Obsessive Compulsive Disorder) are main indications for TMS.
TMS is found to be beneficial in other numerous conditions such as Anxiety, Autism Spectrum Disorders, PTSD, Addiction, ADHD, Eating Disorders and Chronic Pain.
TMS can be combined with psychotherapy, existing antidepressant medications after appropriate CONSULTATION with physician psychiatrist for safety.
Most common side effects of TMS: Headache, more so first couple of initial treatments. Others: prickly, tingly sensations of the scalp while stimulating pulses are being administered. About one third of patients experience this, which is nothing to worry about, and the effects should go away with time. ONE in 1,000 patients treated may experience mild seizure; however, it should be noted that this risk is approximately is the same as that caused by some FDA approved anti-depressant medications. Additionally, the seizures related to TMS typically end when the magnetically stimulation is stopped.
However, patients who have metal implants in or near their head should not have TMS. Exceptions include those with dental fillings, crowns and/or brace, AFTER CONSULTATION with physician-psychiatrist.
Metals that prevent TMS Treatment include: (contraindications)
- Pacemakers or implanted defibrillators
- Any stents from the neck up
- Aneurysm clips
- Metallic implants in ears or eyes
- Ammo fragments near the Head
- Facial Tattoos with metallic Ink
- Any metal near the Head that cannot be removed (Earrings, glasses, facial piercings)
PHONE: (704) 868-8988
ADDRESS: 839 Majestic Ct, Suite 8, Gastonia, NC 28054